There has been increasing interest in extracellular vesicles (the “Exosome”) as possible biomarkers for disease and as novel cell-cell communicators. Extracellular vesicles (EVs) carry a variety of cargo such as mRNA, non-coding RNA, proteins and lipids and are thought to have a functional role in both normal physiology and pathological conditions. They are released from many cell types and circulate in multiple biofluids in the body, including plasma, cerebrospinal fluid, and urine.
Addition of TransFix at the point of blood collection may reduce the negative impact of platelet EV release, giving researchers, a better idea of the events happening in patient samples
TransFix is a fixative designed for stabilizing cell surface antigens until sample processing and analysis can be performed. EVs in the blood are stable even after freeze/thaw cycles. However, plasma processing at 4C, may cause more EVs to be released from platelets. This EV release complicates data generated by scientists looking for rare exosomal biomarker events. The study below, demonstrates that addition of TransFix at the point of blood collection may reduce the negative impact of platelet EV release, giving researchers, a better idea of the events that may be happening in vivo.
Case Study: A Comparison of Extracellular Vesicle Isolation from Blood Collection with Conventional EDTA and TransFix/EDTA Vacuum Blood Collection Tubes
Summary
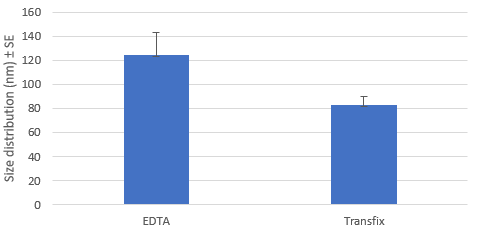
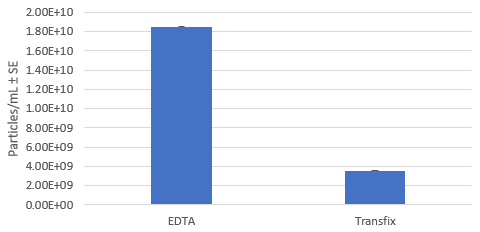
The EV profile (CD9, CD63, and CD81 tetraspanin proteins) of normal serum with and without the use TransFix containing tubes, after storage at 4°C for 4 days was evaluated. Western blot analysis showed detection of all three EV tetraspanin proteins. NanoSight analysis showed that the TransFix tubes were more likely to capture particles of the correct size range for EVs and that the particle concentration is higher for the EDTA samples than the TransFix samples. This higher concentration in the EDTA plasma sample is believed to be due to the release of EV from platelets during processing and storage at 4°C.
Western blot analysis showed detection of all three EV tetraspanin proteins.
NanoSight analysis showed that the TransFix tubes were more likely to capture particles of the correct size range for EVs (1).
The particle concentration is higher for the EDTA samples than the TransFix samples. This higher concentration in the EDTA plasma sample is believed to be due to the release of EVs from platelets during processing and storage at 4°C (2)(3).
The addition of TransFix at the point of blood collection may reduce the negative impact of platelet EV release, providing a more accurate picture of the EV content of samples
Method
On day 0 a blood draw from donors was performed. Each donor provided 3 x 9 ml of blood drawn into conventional EDTA Vacuum Blood Collection Tubes and 3 x 9 ml of blood drawn into the TransFix/EDTA vacuum blood collection tubes, (Product Code TVT-09-50) from a single venipuncture. The tubes were centrifuged at 300 x g for 20 minutes at room temperature. The upper plasma layer was separated and divided into 1 ml aliquots and samples were stored at 4°C. On day 4, exosomes were isolated from the EDTA and TransFix plasma samples (using an ExoCap™ Streptavidin Kit, MBL International). The isolated EVs were then analysed by western blot to visualize EV proteins and by NanoSight to determine the EV size and concentration.
Results
Eluted Extracellular Vesicle Size Distribution: EDTA vs TransFix Tubes
Eluted Extracellular Vesicle Concentration: EDTA vs TransFix Tubes
TransFix Products
 TransFix/EDTA Vacuum Blood Collection Tubes (TVTs) are plastic, direct draw collection tubes used to stabilise venous blood at the point of collection and preserve whole blood specimens for immunophenotyping by flow cytometry. TVTs are prefilled with sufficient TransFix containing K3EDTA for the immediate anticoagulation and stabilisation of 3ml or 9ml of blood for up to 14 days.
TransFix/EDTA Vacuum Blood Collection Tubes (TVTs) are plastic, direct draw collection tubes used to stabilise venous blood at the point of collection and preserve whole blood specimens for immunophenotyping by flow cytometry. TVTs are prefilled with sufficient TransFix containing K3EDTA for the immediate anticoagulation and stabilisation of 3ml or 9ml of blood for up to 14 days.
References
- Graça Raposo, Willem Stoorvogel (2013). Review: Extracellular vesicles: Exosomes, microvesicles, and friends. Journal of Cellular Biology 200 (4): 373
- Heijnen, F.G. et al (1999). Activated Platelets Release Two Types of Membrane Vesicles: Microvesicles by Surface Shedding and Exosomes Derived From Exocytosis of Multivesicular Bodies and Granules. Blood 94; 3791-3799.
- Aatonen, A.T et al (2014). Isolation and characterization of platelet-derived extracellular vesicles. Journal of Extracellular Vesicles. 3 (1); 24692